 |
|
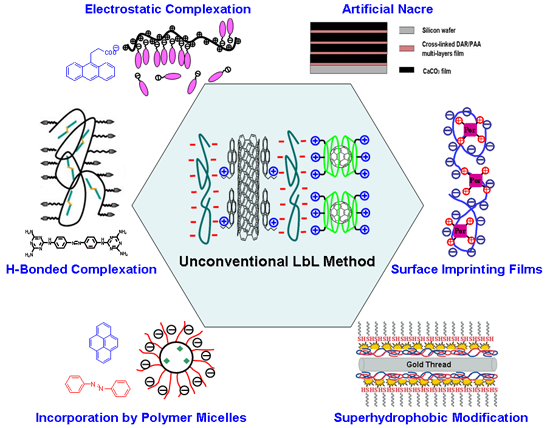
Layer-by-layer (LbL) assembly is a powerful means for fabricating multilayer thin films with controlled architecture and composition. Although many materials have been assembled to construct functional multilayered thin films, there are still some functional species which cannot be assembled using conventional LbL methods. For this purpose, we have developed some unconventional methods which usually involve the formation of supramolecular structures via one type of self-assembly. These structures can subsequently be used as building blocks in another type of self-assembly. To take advantage of these conventional and unconventional methods, a great number of building blocks can be fabricated into multilayer thin films with a defined sequence structure in a designed way. For example, we have used block copolymer micelles as supramolecular containers to incorporate water insoluble molecules, e.g. pyrene, into PSt-b-PAA micellar cores, and then employed the pyrene loaded block copolymer micelles as building blocks for layer-by-layer deposition. Interestingly, the incorporating and releasing of the loaded species can be controlled by ionic strength as well as influenced by the core-shell structures of the block copolymer micelles. In addition, we have also proposed a hierarchical assembling method that involves electrostatic complexation and layer-by-layer deposition for the incorporation of single-charged building blocks. By this method, we can realize the selective adsorption and desorption of the functional molecules in the LbL films, which can be further applied in the area of surface molecular imprinting.
|
|
 |
| Recent Publications: |
| Langmuir 2004, 20, 9366; Langmuir 2004, 20, 11828; Chem. Mater. 2005, 17, 5065; Chem. Mater. 2005, 17, 6679; Langmuir 2006, 22, 3906; Chem. Commun. 2007,1395; Langmuir 2007, 23, 2474; Langmuir 2007, 23, 2474; Langmuir 2007, 23, 6377; Langmuir 2007, 23, 11631; Macromolecules 2007, 40, 653; Chem. Mater. 2007, 19, 1974; Adv. Funct. Mater. 2007, 17, 1821; Langmuir 2008, 24, 11988; Langmuir 2009, 25, 2949; Langmuir 2010, 26, 9736; Adv. Mater. 2010, 22, 2689; Langmuir 2010, 26, 15022; Langmuir 2011, 27, 10370; Small 2012, 8, 517; Chem. Eur. J. 2012, 18, 14968; Langmuir 2013, 29, 6348; Small 2013, 9, 3981; Chem. Commun. 2014, 50, 11173. |
|
| |
 |
|
|
|